Pedicle Screw System by G Surgical
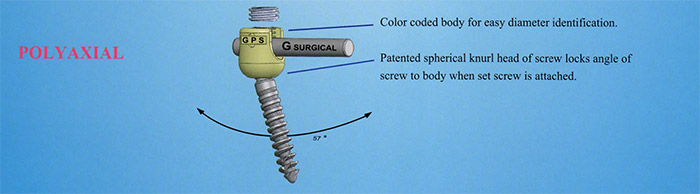
The G Surgical Pedicle Screw System (GPS) is a low profile, top loading, multi-axial system to address pathologies of the thoracolumbar spine. It features 57 degrees of angulation for optimal location placement. GPS utilizes a 6 mm rod design for maximum construct security.
All products are engineered from implant grade titanium, USA manufactured to ISO 13485 requirements, tested at a leading USA University to ASTM standards and FDA cleared.
G Surgical stainless steel instruments feature anatomical silicone handles, are designed for variation in surgical techniques and pathology to assure repeatable and safe surgical outcomes.
Bio-Mechanical Testing
The G Surgical Pedicle Screw System was tested at the bioengineering department of a leading U.S. university per ASTM Fl717-04 protocol, “Standard test methods for spinal implant constructs in a vertebrectomy model.” This comprehensive testing incudes axial compression, static torsion and dynamic compression. Based on positive test results, the G Surgical Pedicle Screw System has been cleared by the FDA for sale in the U.S.
ANAX™ Pedicle Screw System by U&i Corporation
The ANAX™ 5.5 Spinal System is a top-loading multiple component, posterior spinal fixation system which consists of pedicle screws, rods, set screws, connectors, and a transverse (cross) linking mechanism. The ANAX™ 5.5 Spinal System allows surgeons to build a spinal implant construct to stabilize and promote spinal fusion.

Key Features
- Top loading for easy introduction of 5.5 mm diameter rod
- A low-profile polyaxial screw head
- Rounded screw head allowing up to 65degrees of angulation
- Double-lead threaded screw for its fast insertion
- No complication but efficiency self tap of the screw
- No cross threading of the set screw
- Rod available in titanium and cobalt chromium (CoCr)
Indications for Use
- Spondylolisthesis (Grade 3 and 4)
- Degenerative spondylolisthesis with objective evidence of neurological impairment
- Trauma (including fractures, dislocation)
- Spinal stenosis
- Deformities or curvatures (defined as scoliosis, kyphosis or lordosis)
- Tumor
- Pseudoarthrosis
- Failed previous fusion
This product has been cleared by the FDA.
Polarfix™ Facet Screw System by U&i Corporation
Polarfix™ Facet Screw Fixation System is intended to stabilize the spine as an aid to fusion through bilateral immobilization of facet joints.

Key Features
- Hold Facet Screw with screw driver when insert to facet joint through K-wire
- The Washer helps compression of Facet Screw for induce the facet joint fusion
- The spikes of the Washer provide the high stabilization
- Easy insertion with Double lead thread
- Easy insertion without tapping
- Allows for minimally invasive surgery
- Providing ample contacting surface between Washer and bone for compression
Indications for Use
- Spondylolisthesis
- Spondylolysis
- Pseudoarthrosis or failed fusion which are symptomatic
- Trauma including spinal fractures and/or dislocations
This product has been cleared by the FDA.
PEEK PLIF Cage by G Surgical

Key Features
- Minimal implant width and tapered tip facilitate insertion
- Multiple implant sizes and height options to adapt to patient anatomy
- Anatomic shape conforms to the vertebral body
- Large central graft area
- Made from PEEK material for biocompatibility and radiolucency
- Tantalum markers for verification of implant positioning
BN PEEK PLIF Cages by Medyssey

Key Features
- Cages made of PEEK OptimaⓇ
- Straight and lordotic (0°, 4° and 8°) cages
- 8mm to 15mm height
- 25mm, 27mm, 32mm, and 34mm length
- Grooved surface
Medussa® 3D-Printed Porous PLIF Cage by Medyssey


With EBM 3D Printing technology
Key Features
- Cages made of Titanium alloy powder
- High volume porosity capable of supporting tissue ingrowth
- High coefficient of friction that can aid in initial device stability
- Straight and lordotic (0°, 4° and 8°) cages
- 8mm to 16mm height
- 25mm, 27mm, 30mm, and 32mm length
Taurus PEEK TLIF Cages by Medyssey

Key Features
- Cages made of PEEK OptimaⓇ
- Larger surface for enhanced bone graft
- Lordotic 7°
- 7mm to 16mm height & Width 10mm and 12mm
- 25mm, 27mm, 30mm, and 33mm length
- Grooved surface
- Available in Banana type and straight type
Varian™ Expandable Cage by Medyssey

Key Features
- Expandable Cage
- Offering 8 & 10 lordotic angles once expanded
- 2.0-2.5mm lifted up in front once expanded
- Made up of Titanium Alloy
- 9 – 15mm in Height
- 24 – 27mm in Length
- Simplified instrumentation to restore disc height and lordosis
- Grooved surface
Velofix™ PEEK Lumbar Cage by U&i Corporation
The Velofix™ PEEK Lumbar Cage allows restoration of the disc height and natural curvature. Nerve root decompression is also achieved by opening the neural foramen. It provides enhanced design, easy and efficient installation with ergonomic instruments to have effective solution for cervical interbody fusion. The Velofix™ PEEK Lumbar Cage is manufactured from durable PEEK-OPTIMA, providing a wide variety of implant sizes, heights and anoatomical shape ofr varying patient anatomies.

Key Features
- Contact surface with prominent serrations for enhancing implant stability
- Central I-Beam design for preventing deformation during placement
- Large central window for improved loading capacity for bone graft material
- Chamfered, bulleted design on leading edge for easier insertion
- Wide side window for bone ingrowth
- Three radiographic tantalum rod makers for improved fluoroscopic placement
- Threading inserter for securing implant
Indications for Use
- Primary surgery for advanced discopathies or extensive decompressions (laminectomy, facetectomy, foraminotomy, etc.)
- Spondylotic spinal stenosis
- Revision surgery for failed disc operation
- Post-operative instability
- Disc herniation
- Recurrent disc herniation
- Spondyloysis with bone spurring, foraminal narrowing and nerve root impingement
- Chronic and disabling discogenic low back pain
- Spinal pseudarthrosis
This product has been cleared by the FDA.
